Which of the Following Atoms Has the Smallest Atomic Radius
Carbon Nitrogen Oxygen Fluorine. Neon has the smallest atomic radius.
Atomic Radius Trend Periodic Table Chemtalk
Which of the following atoms has the smallest atomic radius.
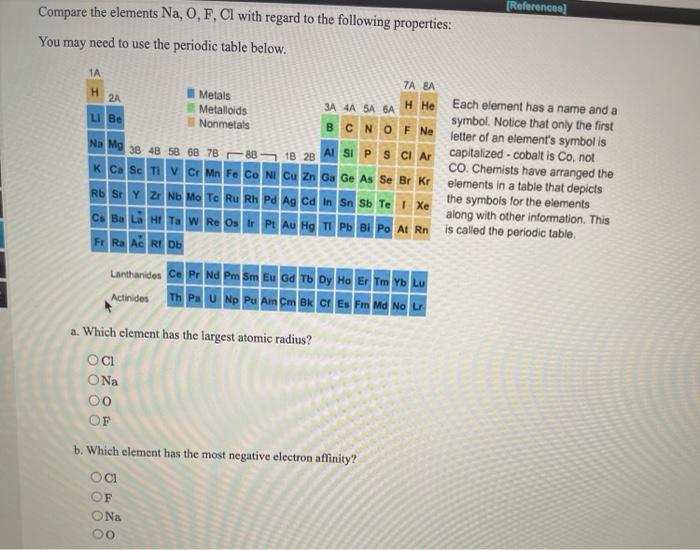
. This is due to trends in the periodic table and the effective nuclear charge that holds the valence electrons close to the nucleus. The concentration of more protons in the nucleus creates a higher effective nuclear charge In other words there is a stronger force of attraction pulling the electrons closer to the nucleus resulting in a smaller atomic radius. So the atom kinds are the same.
Helium has the smallest atomic radius. Does P or S have a larger atomic radius. Rank these elements in terms of increasing atomic radius.
Helium has the smallest atomic radius. Which of the following elements has the smallest atomic radius. The radius of the potassium atom is smaller because of its smaller nuclear charge.
From the given elements potassium has the largest atomic radius. What elements have the smallest atomic radius. Question 10 1 point Put the following atoms in order from largest to smallest atomic radius largest 1 smallest 3.
Phosphorus has a larger atomic radius than Sulfur. Which of the following correctly lists the five atoms in order of the five atoms in order of increasing size smallest to largest. Which of the following atoms has the smallest covalent atomic radius.
Lithium _____ is a phenomenon that may be caused by the burning of fossil fuels which increases the carbon dioxide content in the earths. Which of the following atoms has the smallest atomic radius. Which of the following elements belongs to representative elements.
Math Chemistry Biology Programming Arts History BusinessLanguage Spanish EnglishTipsReviewBlog Home Order the following atoms according increasing atomic radius SFO. As can be seen in the figures below the atomic radius increases from top to bottom in a group and decreases from left to right across a period. To do this we have to assume that they are given the same number of atoms as the atom types.
Which statement best compares the atomic radius of a potassium atom and the atomic radius of a calcium atom. B atom types C atom type We can say that the atom types make the atom types A and B the same. What is one of the following that has the smallest atomic radius Li Ne Rb Sr or Xe.
So all these elements belong to the 4th group and Kr has the smallest atomic radii as it lies across the period. In a period table in group from top to bottom atomic radius increases since the nuclear charge decreases decreases down the group due View the full answer Transcribed image text. Which of the following atoms has the smallest atomic radius.
Of the following which gives the current order for the atomic radius for Mg Na P Si and Ar. A atom type A atom type b. The radius of the potassium atom is smaller because of its larger.
This means the atom types give different numbers of atom types. Thus helium is the smallest element and francium is the largest. Which one of the following atoms has the smallest radius.
Ocmgrbcs or arpalnak -barbie H smallest S Si Na Rb largest. July 25 2021July 25 2021 thanh Rank them.

Solved Of The Following Which Atom Has The Smallest Atomic Chegg Com

Solved A In The Following Set Which Atom Has The Smallest Chegg Com

Periodic Trends Determine Which Atom Has The Smallest Atomic Radii Radius Johnny Cantrell Youtube

No comments for "Which of the Following Atoms Has the Smallest Atomic Radius"
Post a Comment